
Introduction
Red light therapy's application to hearing loss is one of the more speculative uses in the photobiomodulation literature - not without basis, but also not with the same evidence depth as its applications to skin, muscle recovery, or wound healing.
This guide covers what the research has explored, why the mechanism is plausible, where the evidence is genuinely limited, and what realistic expectations look like for someone considering this application.
Key Takeaways
- Near-infrared light therapy (around 808-850nm) has been investigated for cochlear protection and hearing support in animal models and early human studies
- The mechanism involves mitochondrial support in the highly energy-dependent hair cells of the cochlea
- Evidence in human subjects is preliminary; most published studies are small, methodologically varied, and not conclusive
- Red light therapy is not an established treatment for hearing loss and should not be positioned as one
- For users interested in exploring this application, near-infrared wavelengths have more mechanistic relevance than 660nm red light for inner ear applications
Why the Mechanism Is Plausible
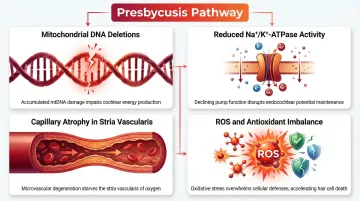
The inner ear - specifically the cochlea - contains hair cells that are among the most metabolically active cells in the body. They are highly dependent on mitochondrial function to maintain the electrochemical gradients required for sound signal transduction.
Cochlear hair cell damage and death are associated with noise-induced hearing loss, age-related hearing loss (presbycusis), and ototoxic drug exposure. Once these cells are lost, they do not regenerate in humans - which is why acquired hearing loss is typically permanent.
Near-infrared light at 808-850nm penetrates tissue more deeply than visible red light (4-8mm versus 1-4mm) and can reach the structures of the inner ear through the temporal bone and surrounding tissue. The proposed mechanism is that mitochondrial support from photobiomodulation could:
- Protect viable cochlear hair cells from ongoing mitochondrial dysfunction
- Support the energy metabolism of remaining auditory neurons
- Reduce inflammatory and oxidative stress in cochlear tissue
This mechanism is supported by animal model research where near-infrared light therapy showed cochlear protective effects in noise-exposure models. The translation to human clinical benefit is less established.
What the Research Shows
Animal Model Studies
Multiple animal studies (primarily in rodent models) have demonstrated:
- Reduced cochlear hair cell loss following noise exposure when near-infrared light was applied pre- or post-exposure
- Preservation of auditory brainstem response (ABR) thresholds in treated versus control animals
- Reduced markers of oxidative stress and inflammation in cochlear tissue
These findings establish that the mechanism is biologically plausible and active in animal models. They do not establish clinical efficacy in humans.
Human Studies
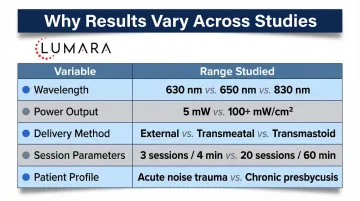
Human studies on red or near-infrared light therapy for hearing are limited and methodologically varied. Key observations:
- Sample sizes are generally small (under 50 participants in most published studies)
- Study designs vary significantly in wavelength, irradiance, treatment duration, and patient population
- Some studies show improvements in audiometric outcomes; others show no significant change
- No large-scale RCT has established near-infrared light therapy as an effective treatment for any form of hearing loss

Tinnitus
Some preliminary research has examined near-infrared light therapy for tinnitus, with variable results. The proposed mechanism differs - tinnitus involves abnormal neural firing rather than hair cell loss - but inflammatory and metabolic factors may overlap. Evidence is similarly preliminary.
Honest Assessment of Where This Stands
Red light therapy for hearing loss is in an early research phase. The mechanism is plausible, animal models are supportive, and some human studies are encouraging. But:
- There is no established protocol for this application
- Published human trials are too small and methodologically varied to draw conclusions
- It is not an approved or clinically established treatment for hearing loss
- It should not be positioned as a treatment for diagnosed hearing conditions
For someone with significant hearing loss, red light therapy is not a substitute for audiological assessment, hearing aids, or medical evaluation of the underlying cause.
Practical Considerations for Users Exploring This
For users who want to explore near-infrared light therapy in the context of auditory wellness:
Wavelength: Near-infrared (808-850nm) has more mechanistic relevance than 660nm red light for inner ear applications. Devices that include near-infrared are more appropriate than red-only devices.
Positioning: Temporal region (the area around the ear and temple) is the most commonly used position in research protocols, allowing near-infrared penetration toward cochlear structures.
Session protocol: Research protocols vary; there is no established standard for home use. Typical research sessions run 10-20 minutes.
Expectations: Any benefit is expected to be protective (reducing further deterioration) rather than restorative (recovering lost hearing). The evidence does not support claims of hearing restoration.
Frequently Asked Questions
Can red light therapy help with hearing loss?
The evidence is preliminary. Near-infrared light therapy has shown cochlear protective effects in animal models and some small human studies. It has not been established as an effective treatment for hearing loss in controlled clinical trials. It is not a substitute for audiological care.
What wavelength is relevant for hearing applications?
Near-infrared at 808-850nm has more mechanistic relevance for inner ear applications than 660nm red light. NIR penetrates more deeply and can reach cochlear structures through temporal bone tissue.
Does red light therapy help with tinnitus?
Preliminary research has examined this question with variable results. The evidence is too limited to draw conclusions. Tinnitus has multiple causes, and light therapy's relevance depends on the underlying mechanism in each case.
Is red light therapy safe near the ear?
Consumer-level near-infrared and red light therapy devices used externally on the temporal region are generally considered low risk. Eye protection should be used during sessions that are near or above eye level. Anyone with active ear infections, implanted cochlear devices, or other otological conditions should consult an ENT before starting.
Where should I position a device for this application?
The temporal region - the area around the ear and temple - is used in most research protocols. This allows near-infrared light to reach the cochlea through the surrounding tissue.
Plausible, Promising, But Still Early
Near-infrared light therapy for hearing support has a coherent mechanistic basis and encouraging early evidence. It is not an established treatment, and anyone with diagnosed hearing loss should prioritize proper audiological care over any light therapy application.
For users interested in exploring near-infrared light therapy more broadly - for recovery, tissue health, and wellness applications with stronger evidence - Lumara's Illuminate V2 at 660nm provides a high-output panel format with verified wavelength delivery for skin and body applications.


