
Introduction
Neuroinflammation - inflammation within the central nervous system - is increasingly recognized as a factor in conditions ranging from traumatic brain injury to neurodegenerative diseases and depression. Near-infrared light therapy applied transcranially has a growing research basis for anti-inflammatory and neuroprotective effects at the brain level.
This is not an established treatment for neurological diseases. But the mechanism is scientifically coherent, early research is encouraging, and understanding what it can and cannot do helps users make informed decisions.
Key Takeaways
- Neuroinflammation involves microglial activation and pro-inflammatory cytokine release that can damage neurons over time
- Near-infrared light at 810-850nm can penetrate through skull and scalp to reach cortical tissue
- The mechanism involves mitochondrial support in neurons, reducing oxidative stress and modulating inflammatory signaling
- Research in TBI, Alzheimer's, Parkinson's, and depression has shown anti-inflammatory effects in animal models and small human trials
- This is early-stage research - transcranial NIR is not FDA cleared for neurological conditions
- Red light therapy is appropriate as a general wellness tool; therapeutic claims for neurological disease should not be made
What Neuroinflammation Is
Neuroinflammation is the brain's inflammatory response to injury, infection, or systemic inflammation. It involves activation of microglia (the brain's resident immune cells) and astrocytes, which release pro-inflammatory cytokines.
Acute neuroinflammation is protective. Chronic neuroinflammation, however, can itself become damaging - contributing to neuron loss in Alzheimer's, Parkinson's, TBI sequelae, and depression.
How Near-Infrared Light Reaches the Brain
Near-infrared wavelengths at 810-850nm penetrate deeper than visible red light. Research has shown that these wavelengths can pass through scalp, skull bone, and meninges to reach the cortical surface.
Penetration is limited at consumer device irradiance - NIR does not reliably reach deep brain structures. It reaches the cortex and upper brain structures most effectively. Deeper brain regions require clinical-grade transcranial devices.
At consumer device levels, transcranial NIR is primarily relevant for cortical applications - the outer brain regions involved in depression, cognitive function, and some aspects of neuroinflammatory conditions.
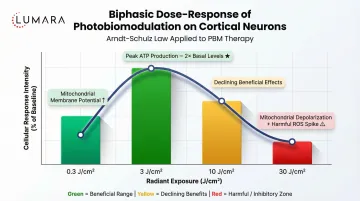
What the Research Shows
Traumatic Brain Injury (TBI): Multiple animal model studies and small human trials show transcranial NIR reducing neuroinflammatory markers, improving cognitive function scores, and reducing microglial activation. Research from Massachusetts General Hospital and other centers has produced positive phase I/II trial data. Evidence is promising but not conclusive from large RCTs.
Neurodegenerative Conditions: Animal model studies in Alzheimer's and Parkinson's contexts show reduced amyloid plaque burden, neuroprotective effects on dopaminergic neurons, and reduced oxidative stress. Human clinical trials are ongoing.
Depression and Cognitive Function: The most advanced human research area. Multiple controlled trials show improvements in depression scales with transcranial NIR applied to the forehead (prefrontal cortex targeting). Evidence is building toward larger trials.
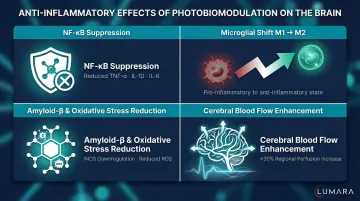
The Mechanism
Near-infrared light in neurons works through photobiomodulation: absorption by cytochrome c oxidase in mitochondria, supporting cellular energy and reducing oxidative stress.
For neuroinflammation: reduced microglial activation and pro-inflammatory cytokine production, neuronal energy support under inflammatory stress, and modulation of inflammatory signaling pathways.
If mitochondrial dysfunction in neurons contributes to chronic neuroinflammation (as research suggests), photobiomodulation-driven mitochondrial support is mechanistically coherent.

Important Limitations
This is not a treatment for neurological disease. Consumer-level NIR devices are not FDA cleared for any neurological condition. The research is at an early stage.
Anyone experiencing neurological symptoms should have a medical evaluation. Red light therapy is most appropriate as a wellness adjunct alongside medical care, not as a primary intervention for diagnosed conditions.
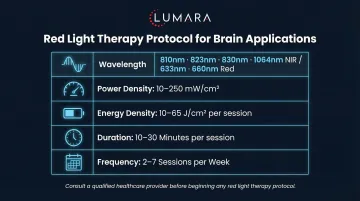
Frequently Asked Questions
Can red light therapy reduce brain inflammation?
Near-infrared light has shown anti-inflammatory effects on neural tissue in animal models and small human trials. It penetrates to cortical tissue through the skull at consumer device levels. This is promising early research - not an established treatment.
What wavelength is used for transcranial light therapy?
Most research uses 810nm or 850nm near-infrared. 660nm red light does not penetrate as effectively to cortical tissue.
Is red light therapy safe for the head?
Consumer-level NIR and red light applied externally to the head is generally considered safe at consumer irradiance levels. Anyone with neurological conditions or concerns about eye exposure should consult a physician first.
Early Research, Real Mechanism, Appropriate Expectations
The research on near-infrared for neuroinflammation is scientifically credible and worth following as evidence matures. It is not a medical treatment, but as a wellness-oriented supportive tool, the mechanistic basis is sound.
For users building a wellness routine with light therapy, Lumara's Illuminate V2 provides verified 660nm output - FDA cleared, consistent irradiance, 5-minute sessions.


