Introduction
Repeated low-level red light therapy (RLRL) has become one of the more closely studied approaches in pediatric myopia control. Unlike pharmaceutical interventions or contact lens methods, it is non-invasive, well-tolerated in children, and has shown meaningful reductions in axial elongation in multiple controlled trials.
This guide covers the clinical basis for RLRL therapy, what the key trials found, the safety profile, and how it compares to other approaches. It is written for parents, clinicians, and serious buyers who want to understand the evidence before making a decision.
Key Takeaways
- RLRL therapy uses 650-660nm red light delivered in short daily sessions to slow axial elongation in myopic children
- Multiple randomized controlled trials have shown meaningful reductions in axial elongation and myopia progression versus control groups
- The mechanism involves choroidal thickening and improved oxygenation to the sclera
- Safety monitoring is required - retinal examinations before and during treatment are standard in clinical protocols
- Device quality matters significantly: wavelength accuracy, output consistency, and session format all affect whether a protocol can be reliably replicated at home
- RLRL is not a cure for myopia; it is a progression management tool used alongside regular eye examinations
What Is RLRL Therapy and How Does It Work
RLRL therapy delivers red light - typically at 650-660nm - to both eyes for short, structured daily sessions. The treatment is used to slow the axial elongation of the eye that drives myopia progression in children.
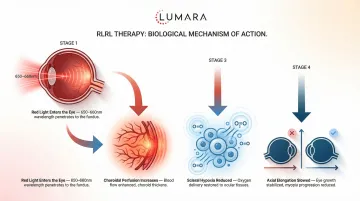
The proposed mechanism centers on two effects. First, red light at these wavelengths appears to stimulate choroidal thickening, which improves oxygenation to the sclera - the outer layer of the eye. Chronic scleral hypoxia (insufficient oxygen supply) is associated with the structural changes that lead to axial elongation. Second, 650-660nm wavelengths interact with photoreceptors and mitochondria in retinal cells in ways that may support normal cellular function.
The key point for buyers and clinicians: wavelength precision matters. The clinical trials that have demonstrated efficacy used devices operating at 650-660nm. Devices that approximate this range or operate at different wavelengths cannot be assumed to replicate these results.
What the Research Shows
Jiang et al. (2022) - Foundational RCT
One of the early well-controlled trials showed that children receiving RLRL therapy had significantly less axial elongation compared to a control group over 12 months. The trial used a 650nm device with two 3-minute sessions per day. Choroidal thickening was observed as an early indicator of response.
Dong et al. (2023) - Efficacy and Predictors
A follow-up trial reinforced the axial elongation findings and identified choroidal thickness change as an early predictor of treatment response. Children whose choroids thickened in early sessions showed better longer-term outcomes, which has implications for monitoring protocols.
He et al. (2023) - Comparative Efficacy
This trial compared RLRL therapy against other myopia control interventions and found RLRL produced comparable or superior results on axial elongation endpoints. The trial also contributed safety data showing no significant adverse events in the treatment group under supervised protocols.
Zaabaar et al. (2024) - Meta-Analysis
A systematic review and meta-analysis pooling data from multiple RLRL trials confirmed a meaningful reduction in myopia progression versus control, with acceptable safety profiles in supervised settings. The authors noted the need for longer follow-up data and standardized protocols across trials.
RLRL in High Myopia
Liu et al. (2025) examined RLRL therapy specifically in children with high myopia (over -6.00D), a population where progression risks and complications are more significant. The trial found reductions in axial elongation in the treatment group, with choroidal thickening again appearing as an early response marker.
Importantly, retinal structural changes were monitored closely. No significant adverse retinal effects were observed in the treated group under the supervised protocol, though the authors emphasized ongoing monitoring as standard practice for this population.
Safety Profile
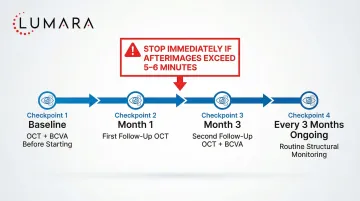
RLRL therapy has a favorable safety profile in supervised clinical settings, but it is not without protocol requirements.
Standard safety practices include:
- Baseline retinal examination before starting treatment
- Regular follow-up examinations (typically every 3-6 months)
- Session time limits (usually 3 minutes per eye, twice daily)
- Output verification of the device being used
- Exclusion of children with pre-existing retinal pathology
The combination of RLRL therapy with atropine has not been studied sufficiently to establish safety, and most clinical protocols treat them as separate interventions rather than combining them. Sessions should follow the recommended eye protection guidance covered in red light therapy eye safety protocols.
Side effects reported in trials have been mild and transient - most commonly brief afterimages following sessions. No serious adverse events have been confirmed in well-supervised protocols.
How RLRL Compares to Other Myopia Control Approaches
| Approach | Mechanism | Typical Efficacy | Key Limitation |
|---|---|---|---|
| RLRL therapy | Choroidal thickening, scleral oxygenation | Meaningful axial elongation reduction in RCTs | Requires supervised protocol, device quality varies |
| Low-dose atropine | Muscarinic receptor blockade | Well-established, titratable | Rebound on discontinuation, systemic considerations |
| Orthokeratology | Corneal reshaping via contact lens | Strong axial control, daily correction | Requires fitting, compliance, infection risk |
| Multifocal soft lenses | Peripheral defocus | Moderate axial control | Compliance, cost |
RLRL therapy's main differentiator is its non-invasive format and tolerability in children who may not adapt well to contact lenses or pharmaceutical protocols. Its limitation is that device quality and protocol adherence are harder to standardize in home settings than in clinical trials.
Device Quality: Why It Matters for Home Use
The clinical trials that established RLRL efficacy used calibrated devices with verified 650-660nm output, controlled session times, and defined output parameters. Replicating those results at home requires a device that can actually deliver the same wavelength accurately and consistently.
This is where the choice of device matters. Lower-cost devices may list 660nm as their wavelength without verifying it at the LED level. Output consistency can drift over time. Session format may not align with clinical protocols. For those considering a DIY red light panel, the same wavelength verification challenges apply.
Lumara's Illuminate V2 is a 660nm red light panel with verified wavelength accuracy, 1,800 LEDs across a 1,200 cm² treatment area, and a 5-minute session format. While it is not a medical device specifically cleared for myopia treatment, it is built around the same wavelength precision that makes RLRL protocols clinically meaningful. For users who want a high-quality 660nm panel for structured daily use, it is one of the more rigorously specified consumer options available.
| Spec | Detail |
|---|---|
| Wavelength | 660nm red light |
| LEDs | 1,800 |
| Treatment area | 1,200 cm² |
| Session time | 5 minutes |
| Energy delivery | 6,000 joules per session |
| Build | Splash-safe |
| FDA status | FDA cleared |
| Made in | USA |
| Eligible for | HSA/FSA |
| Warranty | 3 years |

What RLRL Therapy Cannot Do
RLRL therapy is a progression management tool, not a treatment that reverses existing myopia. It does not reduce current refractive error. It is used to slow the rate at which myopia worsens, particularly during childhood and adolescence when progression is fastest.
It is also not a substitute for regular eye examinations. Children using RLRL therapy in any setting - clinical or home - should have baseline and follow-up retinal examinations as part of their protocol.
Frequently Asked Questions
What is repeated low-level red light therapy (RLRL) for myopia?
RLRL therapy is a non-invasive approach to myopia control that uses low-power 650-660nm red light delivered in short daily sessions. Multiple randomized controlled trials have shown it can slow axial elongation - the primary structural driver of myopia progression - in children.
How does RLRL therapy work?
The main proposed mechanism is choroidal thickening. Red light at 650-660nm appears to increase choroidal blood flow and thickness, which improves oxygenation to the sclera. Chronic scleral hypoxia is associated with the axial elongation that drives myopia. Improved oxygenation may slow this process.
Is RLRL therapy safe for children?
Clinical trials have reported a favorable safety profile under supervised protocols. Standard practice includes baseline and follow-up retinal examinations, controlled session times (typically 3 minutes per eye, twice daily), and exclusion of children with pre-existing retinal conditions. Mild transient afterimages are the most commonly reported side effect.
How effective is RLRL therapy compared to other myopia control methods?
Trials have shown RLRL therapy produces meaningful reductions in axial elongation versus control. Comparative trials suggest efficacy comparable to or exceeding some other approaches. Its advantage is non-invasive format and tolerability in children who are not suitable for contact lens or pharmaceutical interventions.
What wavelength does RLRL therapy use?
Clinical trials have used devices operating at 650-660nm. Wavelength accuracy is important - devices that approximate this range without verified output cannot be assumed to replicate trial results.
Can RLRL therapy be done at home?
Home use is possible with an appropriate device, but it requires adherence to the same session protocol used in clinical trials and should be done under the guidance of an eye care professional who can conduct baseline and follow-up retinal examinations.
Does RLRL therapy stop myopia or reverse it?
No. RLRL therapy is a progression management tool. It slows the rate at which myopia worsens during the treatment period; it does not reduce existing refractive error or permanently halt progression. Regular monitoring is essential.
The Evidence Supports It - The Protocol Makes the Difference
RLRL therapy has a solid and growing evidence base for myopia control in children. The key variables that determine whether those results can be replicated - outside of a clinical trial - are protocol adherence, regular retinal monitoring, and device wavelength accuracy. For a broader look at how red light interacts with mitochondria and cellular function, as well as the latest red light therapy clinical trial findings, those resources provide useful context.
Lumara's Illuminate V2 is built around the 660nm wavelength precision that makes RLRL clinically meaningful - 1,800 LEDs, verified output, 5-minute sessions, FDA cleared, Made in the USA.