
Introduction
Red light therapy research has expanded significantly over the past decade. Controlled clinical trials now cover a range of applications - from skin collagen and wound healing to neurological conditions and metabolic health. Understanding what the research shows, and where it is still early-stage, helps buyers make more informed device decisions.
This guide covers the most clinically relevant research areas, explains what current trial findings mean practically, and distinguishes between strong evidence and emerging work.
Key Takeaways
- Red light therapy has well-established clinical evidence for wound healing, oral wound healing, skin quality and collagen, and muscle recovery
- Emerging research areas include Parkinson's disease, myopia control, traumatic brain injury, depression, and metabolic conditions
- Evidence quality varies significantly by application - some areas have multiple RCTs; others have primarily animal models and small pilot studies
- The wavelength used in a trial matters: consumer devices that approximate rather than verify wavelength may not replicate research conditions
- For buyers, the practical implication is: choose applications where evidence is strong, and choose devices with verified wavelength output that matches the research
Strong Evidence Areas
Skin Collagen and Texture
Multiple randomized controlled trials demonstrate that consistent 660nm red light therapy produces measurable increases in collagen density and improvements in skin texture and firmness. This is among the most-replicated finding in consumer-accessible photobiomodulation research. The mechanism (fibroblast stimulation via mitochondrial photobiomodulation) is well-documented.
What this means for buyers: 660nm red light for facial skincare is one of the most evidence-supported at-home applications. Device wavelength accuracy at 660nm is critical - verified output produces more reliable results than approximate output.
Wound Healing and Oral Healing
One of the earliest and most well-documented photobiomodulation applications. Clinical trials in dental settings show reduced healing time for oral wounds, canker sores, and post-extraction sites. Broader wound healing research shows consistent benefits for surface wound closure and tissue repair.
What this means for buyers: Post-surgical oral applications (canker sores, post-procedure tissue) are among the most evidence-supported consumer applications. 630-670nm wavelength is relevant here.
Muscle Recovery and DOMS
Multiple controlled trials show near-infrared and red light therapy reducing delayed onset muscle soreness (DOMS), improving recovery time, and supporting strength recovery after strenuous exercise. Both 660nm and 850nm wavelengths are represented in this literature.
What this means for buyers: Body-use light therapy for recovery has strong support. Near-infrared (810-850nm) is relevant for deeper muscle tissue; red (660nm) for surface anti-inflammatory effects.

Emerging Research Areas
Parkinson's Disease
Research on transcranial and intranasal near-infrared photobiomodulation for Parkinson's is advancing. A 2023 trial from Australia showed measurable improvements in motor and non-motor symptoms in treated patients versus controls over 12 months. This is early-stage but meaningful - one of the first credible RCTs in a serious neurodegenerative condition.
Important caveat: This research uses clinical-grade devices at higher irradiance than consumer devices. Consumer device irradiance is lower; results may not directly replicate. Do not interpret this research as confirmation that consumer panels treat Parkinson's. Related: red light therapy and brain inflammation.
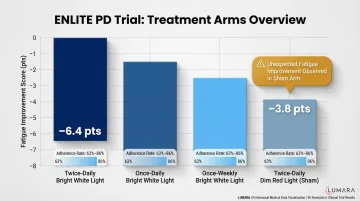
Myopia Control
Repeated low-level red light (RLRL) therapy for myopia control in children has multiple published RCTs showing meaningful reductions in axial elongation versus control groups. This is now one of the more robustly evidenced niche applications of photobiomodulation. The devices used in myopia research operate at 650-660nm applied directly to both eyes for short daily sessions.
What this means for buyers: Myopia control via RLRL is among the more credible emerging applications with multiple RCTs. The specific protocol and irradiance matter; this is not a general "use any red light device" application.
Traumatic Brain Injury
Transcranial near-infrared therapy for TBI has produced positive phase I/II results in controlled studies. Motor recovery, cognitive function, and neuroinflammatory markers have shown improvements. Larger RCTs are ongoing.
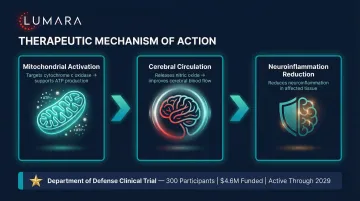
Depression and Cognitive Performance
Transcranial NIR applied to the prefrontal cortex has multiple controlled human trials showing improvements in depression symptoms and cognitive measures. This is the most developed neurological application for consumer-accessible light therapy.
Metabolic Health
Research on photobiomodulation for insulin sensitivity, adipose tissue metabolism, and thyroid function is active. Results are preliminary; this area has fewer high-quality human RCTs than the applications above.
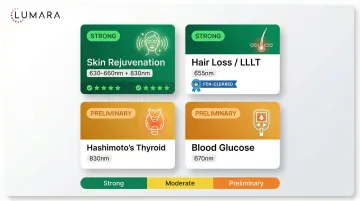
What the Research Means for Device Selection
The clinical trials that produce strong evidence have one thing in common: they use verified wavelength devices at documented irradiance levels. The evidence does not transfer to generic "red light" devices that do not verify their output.
Key buying implication: The research supports specific wavelengths (660nm for skin/wound/muscle, 810-850nm for deep tissue/neurological). Devices that verify output at these wavelengths are more likely to produce outcomes consistent with the research.
For skin applications specifically, 660nm is the most consistently supported wavelength across the broadest range of trials. This is why Lumara's approach of triple-testing wavelength output matters in the context of clinical evidence - verified 660nm is different from claimed 660nm.
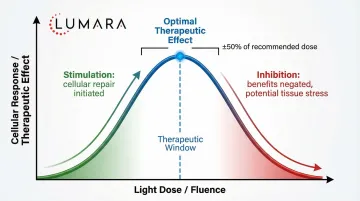
Frequently Asked Questions
What is red light therapy clinically proven to do?
The strongest clinical evidence is for wound healing, oral wound healing, skin collagen and texture improvement, and muscle recovery. These areas have multiple RCTs with consistent results. For skin applications, rosacea is an area where evidence is developing alongside the stronger collagen and texture findings.
What are the most promising emerging research areas?
Myopia control (RLRL), Parkinson's disease, depression (transcranial NIR), and traumatic brain injury. These have early or phase I/II RCT evidence; larger trials are ongoing.
Can I replicate clinical trial results with a consumer device?
For well-supported applications (skin collagen, muscle recovery), quality consumer devices with verified wavelength output can replicate the conditions that produce research results. For neurological applications, clinical devices operate at higher irradiance than consumer devices; direct replication is less certain.
Why does wavelength accuracy matter so much?
Clinical trials specify wavelengths because the mechanism is wavelength-specific. A device that outputs 640nm instead of 660nm is not delivering the same stimulus. Third-party wavelength verification is the only way to confirm a device matches research conditions.
Evidence-Backed Wavelength. Purpose-Built Device.
The clinical research points consistently at 660nm for skin and soft tissue applications - the most accessible and well-supported consumer photobiomodulation application. A device that verifies its output at this wavelength is one that aligns with the research behind it.
Lumara's Illuminate V2 - 660nm triple-verified, 1,800 LEDs, FDA cleared, 5-minute sessions - is built around the wavelength specification that the evidence supports.


