
Introduction
Retinitis pigmentosa (RP) is an inherited progressive retinal dystrophy causing gradual photoreceptor loss, typically beginning with peripheral vision and eventually affecting central vision. No curative treatment currently exists. Management focuses on slowing progression and preserving remaining function.
Near-infrared and red light therapy has generated scientific interest as a neuroprotective approach for RP, with some published research suggesting potential benefits for photoreceptor preservation. This is not an established treatment - it is active early-stage research with encouraging signals. Unlike heat lamp therapy, photobiomodulation operates through photochemical mechanisms at precise wavelengths rather than thermal effects.
Key Takeaways
- RP involves progressive degeneration of rod and cone photoreceptors driven by genetic mutations affecting the phototransduction cascade or supporting cells
- Near-infrared light (670nm and 810-850nm) has shown neuroprotective effects on photoreceptors in animal models and some early human studies
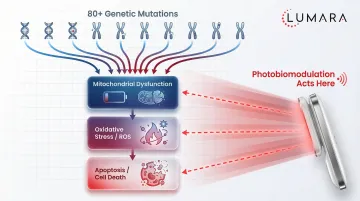
- The mechanism involves mitochondrial support in highly energy-demanding photoreceptors - the same photobiomodulation pathway as other applications
- Results in human studies are preliminary; larger controlled trials are needed before clinical recommendations can be made
- Any RP management should be under ophthalmologist supervision - consumer devices are not ophthalmic devices and cannot replace medical care
What RP Is
Retinitis pigmentosa refers to a group of genetic disorders affecting the retina. The mutations disrupt normal photoreceptor function and lead to progressive cell death, typically:
- Rod photoreceptors (peripheral and low-light vision) degrade first
- Cone photoreceptors (central and color vision) may degrade later
The progression rate varies by genetic subtype. Many people with RP maintain functional vision for decades; others progress to legal blindness relatively early.

The Research Basis for Photobiomodulation in RP
Why Photoreceptors Are Susceptible to Mitochondrial Intervention
Photoreceptors, particularly cones, have some of the highest metabolic demands of any cells in the body. Their function in light transduction is extraordinarily energy-intensive. Mitochondrial dysfunction is a proposed contributor to photoreceptor vulnerability in RP.
This makes photobiomodulation mechanistically plausible for RP: if near-infrared light can support mitochondrial function in photoreceptors, it may reduce the rate of energy-related cell death.
Animal Model Research
Multiple animal model studies (primarily using rodent RP models) have shown:
- Reduced photoreceptor loss in light-treated versus control eyes
- Preserved electroretinogram (ERG) responses indicating retained photoreceptor function
- Effects at both 670nm red and 810-850nm near-infrared wavelengths
Early Human Research
Human research in RP is limited but has produced some positive signals:
- A pilot study using 670nm red light applied transcranially showed improvements in some visual function measures in RP patients
- Research groups at institutions including University College London have published positive preliminary findings
These are small-scale studies with limited follow-up. They are encouraging but not definitive.
Critical Limitations
- No large-scale RCT has established photobiomodulation as an effective treatment for RP
- Most positive research is in animal models, which do not always translate to human outcomes
- Consumer devices operate at lower irradiance than research devices
- The genetic heterogeneity of RP means results may vary significantly by subtype

Appropriate Context for Patients
Red light therapy for RP is appropriately understood as:
- A neuroprotective research area with real but early evidence
- A potential complement to other RP management approaches, not a standalone treatment
- An area where patients should work with their ophthalmologist or retinal specialist to understand current trial opportunities and clinical guidance
If you have RP and are interested in photobiomodulation, the most appropriate next step is discussing it with your retinal specialist, who can assess whether current clinical trials or supervised light therapy protocols are appropriate for your specific RP subtype and disease stage.
Consumer facial LED devices and body panels are not ophthalmic devices. They are not designed or evaluated for therapeutic ocular applications. Do not attempt DIY ocular light therapy with consumer wellness products. Questions about whether eyes should be open or closed during light therapy sessions are separate from the ocular treatment protocols discussed in RP research - always follow clinical guidance.

Frequently Asked Questions
Can red light therapy help retinitis pigmentosa?
Near-infrared and red light therapy has shown neuroprotective effects in animal RP models and some early human pilot studies. It is not an established treatment. Research is active; speak with your ophthalmologist about current evidence and clinical trial opportunities.
What wavelength is used in RP research?
Most research uses 670nm red light or 810-850nm near-infrared. These are different from or at the edge of standard consumer device wavelengths.
Should I try consumer devices for retinitis pigmentosa?
Consumer wellness devices are not designed for therapeutic ocular applications. If you are considering light therapy for RP, this should be through a supervised clinical or research protocol under ophthalmologist guidance.
Is there any approved treatment for retinitis pigmentosa?
Gene therapy (voretigene neparvovec, approved for a specific RPE65 mutation subtype) is the only FDA-approved treatment for a specific RP mutation. Most patients with other subtypes do not have approved treatments; management focuses on supportive care, visual aids, and protecting remaining function.
Early Research, Real Mechanism, Medical Supervision Required
The research on photobiomodulation for RP is scientifically grounded and worth watching as it develops. It is genuinely encouraging science - but not ready for consumer device self-treatment.
For users building general wellness routines with light therapy, Lumara's Illuminate V2 provides precision 660nm output for skin and body wellness applications where the evidence is well-established.


